A Review and Update on the Ophthalmic Implications of Susac Syndrome
- Review
- Open Access
- Published:
Susac syndrome (Retino-cochleo-cerebral vasculitis), the ophthalmologist in the office of the whistleblower
Journal of Ophthalmic Inflammation and Infection book ten, Article number:27 (2020) Cite this commodity
Abstract
Background/purpose
Susac syndrome is a rare microangiopathy of suspected autoimmune origin affecting arteries of the retina, the cochlea and the brain. The aim of the study was to give a review of the disease entity and determine the proportion of cases and their characteristics in a uveitis referral eye.
Patients and methods
Charts of patients with the diagnosis of Susac syndrome seen in the Uveitis Clinic of the Centre for Ophthalmic Specialised Care (COS), Lausanne, Switzerland were reviewed retrospectively to determine the frequency of such cases in a uveitis referral heart. Clinical symptoms and signs, functional information, imaging signs and evolution were analysed in the three COS cases and i case shared with the Uveitis Clinic of the Department of Ophthalmology, University of Innsbruck, Austria. Feature signs were searched possibly allowing a prompt diagnosis.
Results
During the flow from 1994 to 2019 (24 years, 2045 patients), 3 charts with the diagnosis of Susac syndrome were found (0.15%). The whole collective, including the additional example, comprised 3 women aged 28, 32 and 63 at presentation and 1 man, anile 42. None of the 3 cases that were referred were diagnosed beforehand. The characteristic detail establish in all 4 cases was the abrupt arterial stop or segmental interruption of arteries and increased staining of arterial wall on angiography more conspicuously shown on indocyanine green angiography that can potentially be proposed as a crucial diagnostic chemical element. All iv cases responded to dual steroidal and non-steroidal immunosuppression. Under treatment, all 4 patients did not show any further development.
Conclusion
Susac syndrome is a multilocation arteritis of the head that can involve the middle, ear and brain often beginning diagnosed by the ophthalmologist. The diagnosis is speedily reached in uveitis referral centres but seems to be missed otherwise, A helpful angiographic sign to exist searched is an precipitous or segmental arterial stop and increased staining of the arterial wall more than clearly seen on indocyanine green angiography. Patients frequently nowadays get-go to the ophthalmologist who should be acting as a whistleblower to avoid severe involvement of the brain.
Introduction, groundwork and aim of study
Susac syndrome (SS) is a rare occlusive microangiopathy (vasculitis) of unknown aetiology and mechanism involving arteries of the retina, cochlea and encephalon [1]. The syndrome is named after John O. Susac who was the first who described the disease in 1979 [2, 3]. It is characterized by a clinical triad of visual disturbances due to branch retinal avenue occlusion (BRAO), hearing loss and encephalopathy. The verbal prevalence is unknown but up to date slightly more than than 300 cases have been published worldwide [4]. It is presumed to exist an autoimmune-mediated endotheliopathy affecting the vessels of the retina, the cochlea and the brain causing ischemic infarcts in these organs [2]. These microinfarcts are leading to the typical clinical triad [5]. Recently anti-endothelial prison cell antibodies (AECA) were detected in 25% of the patients supporting the hypothesis of an autoimmunity targeting the microvasculature [half-dozen, seven]. It has been shown recently that CD8+ T cell-mediated endotheliopathy is the mechanism of arterial wall inflammation in Susac syndrome that tin can exist blocked by anti-α4 integrin monoclonal antibodies [8]. Eyes obtained at dissection from patients with SS confirmed in histopathological exam artery occlusion at side of endothelial prison cell dysfunction and glia besides seems to be involved [9]. The blood vessels often lacked viable endothelial cells, the wall of the arteries appeared thickened with amorphous material and dome shaped serous similar material was located below the internal limiting membrane [9].
Clinical presentation
Ophthalmic findings
At least 50% of patients have visual disturbances as first clinical manifestation [4]. Patients complain almost reduced visual acuity, scintillating scotomas, photopsia or visual field defects. The characteristic fundoscopic findings in patients with SS are branch retinal artery occlusion or arterial narrowing and small punctuate xanthous-white arterial wall plaques; these plaques are also called Gass plaques [10] and can resolve overtime [10, 11]. The findings in retinal fluorescein angiography (FA) are pathognomonic and bear witness segmental arteriolar wall hyperfluorescence (AWH) with dye leakage in 96% of the patients [four], ofttimes occurring in a multifocal manner and located distant to areas of branch retinal avenue occlusion (BRAO). Moreover, non-perfused retinal arterioles or arterial luminal narrowing with a preserved downstream blood perfusion tin can be institute in FA. This arterial mural staining indicating an impaired integrity of the arterial or arteriolar wall may be found unilaterally or bilaterally [x]. A progression of the AWH into BRAO has been documented in some cases but it is unclear why some AWH result in BRAO and others do not. It is important to know that AWH and arterial luminal narrowing in FA can even be found in a normal appearing fundus [12]. Indocyanine greenish angiography (ICGA) is showing hypofluorescence in the areas of retinal infarction and is also showing retinal vessel aberration while choroidal circulation appears as normal [thirteen]. Optical coherence tomography (OCT) has recently become a valuable diagnostic tool. In a case series, 68% of SS optics showed significantly reduced average retinal nerve fibre layer thickness (RNFLT)). Characteristic is the very distinct pattern of patchy thinning of the inner retina while the outer retina remains normal reflecting arterial distribution [14]. In OCT sectors with severe inner retinal thinning are located side by side to normal appearing sectors [12, fourteen]. Oct provides complementary diagnostic data to FA especially in chronic or afterwards stages of the disease.
CNS manifestation
The well-nigh mutual clinical manifestation at onset of SS is encephalopathy (ii thirds of patients) [iv]. The symptoms are headache, cognitive harm, changes in personality, sensory and motor disturbances, clutter and confusion. On MRI involvement of the corpus callosum with typical small multifocal snowball-like lesions in T2 weighted images tin be found in 78% in the acute phase and are considered as a characteristic sign of SS [15]. Moreover T2 weighted images show supratentorial white affair lesions in 98% and T2 or FLAIR hyperintense lesions in 70–100% in the periventricular white affair, subcortically, and in the deep grey matter nuclei [15, 16]. The central callosal lesions differ from those in demyelinating disease, which is the most important differential diagnoses. The cerebrospinal fluid shows a moderate elevation of proteins and a balmy pleiocytosis. Oligoclonal bands (OCBs) can exist establish in about 15% of the patients differentiating SS from MS where oligoclonal OCBs can be constitute in up to 98%, being helpful just when OCBs are negative [17].
Hearing damage
Hearing loss can occur overnight involving one ear with the 2d ear following inside a few days. A loss of low or midtone range is typical merely also a loss of high frequencies tin be observed. Roaring tinnitus and vertigo are frequently accompanied with hearing loss or can precede it [i,2,3,4].
Clinical course
The typical age at onset of the disease is between 20 and 40 years of age but the age range extends from 2.5 to 72 years [4, xviii, 19]. It predominantly affects women with an estimated male/female ratio of about one:iii.
Three clinical courses tin can be distinguished: A monophasic, a polyphasic and a chronic continuous grade. In the majority of cases (54%) SS is monophasic, predominantly with encephalopathy and oft express to 1–2 years with a skilful prognosis if treated early [18]. In the polyphasic course patients suffer from recurrent branch retinal artery apoplexy and hearing loss over several years. The time between the relapses tin exist very long upwardly to eighteen years [19]. In the chronic continuous grade, symptoms are fluctuating without real periods of remission.
Differential diagnosis
The almost probable differential diagnoses are inflammatory demyelinating CNS diseases similar multiple sclerosis (MS) and acute disseminating encephalomyelitis (ADEM). Other important differential diagnoses are retinal vasculitis with or without systemic affliction [20].
Treatment
As SS is a rare disease no randomized controlled trials take been published and handling strategies vary considerable and are based on the results of case series. Based on the hypothesis of being an autoimmune illness treatment has to be immunosuppressive. As start line high dose iv. corticosteroids are recommended with methyprednisolone (500 - 1000 mg/d) for 3 days followed by oral dose of 1 mg/kg per day for the start 2 to 4 weeks; tapering is depending on the clinical picture and tin can be between 10 and xx% every two weeks [21,22,23,24]. To reduce corticosteroids immunosuppressive agents similar mycophenolate mofetil, azathioprine or cyclosporine, should exist added early [22, 25]. Other possible treatment options are plasma exchange or the application of subcutaneous immunoglobulins (sc IgG) [26]. Treatment with monoclonal antibodies (Rituximab) or tumor necrosis factor (TNF) inhibitor Infliximab take been described [27, 28]. In addition to reduce the risk of microvascular thrombosis and vascular occlusion, treatment with antiplatelet agents and antivasosplastic agents can exist considered [2, 24].
Aim
The aim of this report was to analyse the ophthalmic clinical and imaging signs of SS in four patients seen in an ophthalmology referral centre. Characteristic features were sought that, when present, would raise the awareness of the ophthalmologist to the diagnosis of SS and and then avoid to, mistakenly, consider these cases as common retinal vasculitis and/or ischemic events. The ophthalmologist is brought to play the role of a whistleblower in this condition in society to avoid potential astringent consequences for the ear and/or the brain.
Results
Frequency
Amid the 2045 new cases of uveitis seen at the uveitis dispensary of the Centre for Ophthalmic Specialised care (COS), Lausanne, Switzerland, 3 patients were diagnosed as Susac syndrome, amounting to 0.15% of cases in a specialised uveitis centre.
Demographics and diagnosis
The series from 2 centres analysed clinically was equanimous of iii women aged 28, 32 and 63 years and 1 human being aged 42 years. 1 patient directly consulted the uveitis referral centre in Innsbruck and was diagnosed without delay. One patient presented a posterior pole retinal infarction with a central scotoma and was immediately referred and diagnosed without delay. For the other ii cases the diagnostic delay was four and 48 months once they were seen in the referral centre (Table i).
Ophthalmic and systemic presenting signs
The abiding ophthalmic presenting sign was a subjective scotoma that was scintillating in ii cases. This was associated with hearing disturbances in 3 cases and cerebral signs such every bit memory loss in two cases. Two patients were under low dose systemic corticosteroid treatment, when seen in our centre.
Ophthalmic features
Snellen nautical chart visual acuity was 1.0 OU in the 2 patients diagnosed early on and in the patient in a subacute phase. In the patient seen 48 months after the initial symptoms, VA was 0.ii OD and 0.9 OS.
There was no anterior uveitis recorded in any of the patients and light amplification by stimulated emission of radiation flare photometry was normal in the two patients with initial onset affliction who underwent this test.
Fundus
Fundus findings depended on the stage of evolution of the disease. The case seen immediately after retinal infarction had been detected by the treating ophthalmologist and showed the typical yellow-white retinal discoloration of the ischaemic zone.
This surface area had disappeared half-dozen weeks later and was replaced by retinal atrophy well shown on optical coherence tomography (OCT). (Fig. 1).
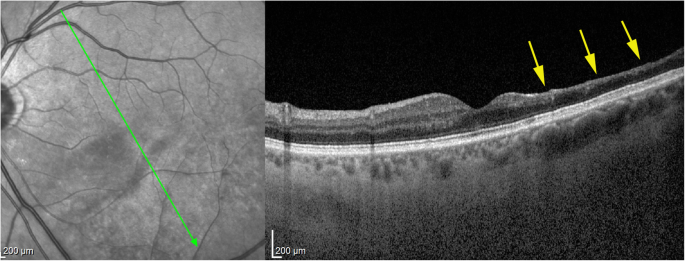
Six weeks after presentation the ischaemic zone resulted in atrophy the of the inner retina; shown past Oct imaging (arrow); notation that photoreceptor outer segment ellipsoid zone is conserved (case 1)
For the other three patients where disease had evolved for more than half dozen weeks no fundus signs of ischaemia could be detected. However, in ii cases segments of white-yellowish discoloured arteries were detected. (Fig. two).
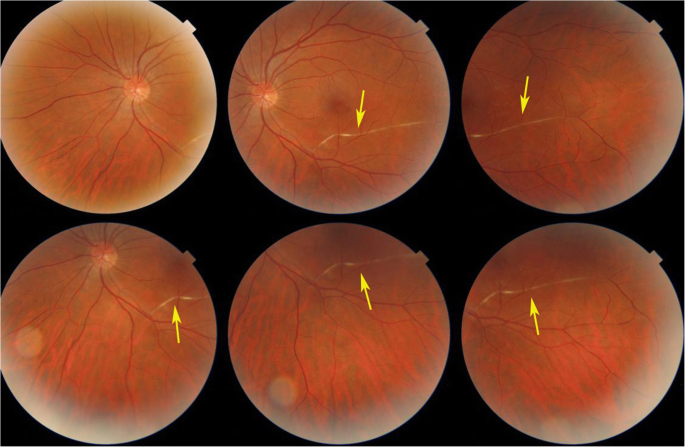
White-yellow discoloration of the course of a branch artery from junior arcade (arrows) in a case evolving since 4 months (instance 3)
Optical coherence tomography (OCT)
In the patient seen in the astute phase (patient one), oedematous thickening of the ischaemic area could be seen at presentation (Fig. 3) evolving towards atrophic thinning of the inner retina. (Fig. 1).
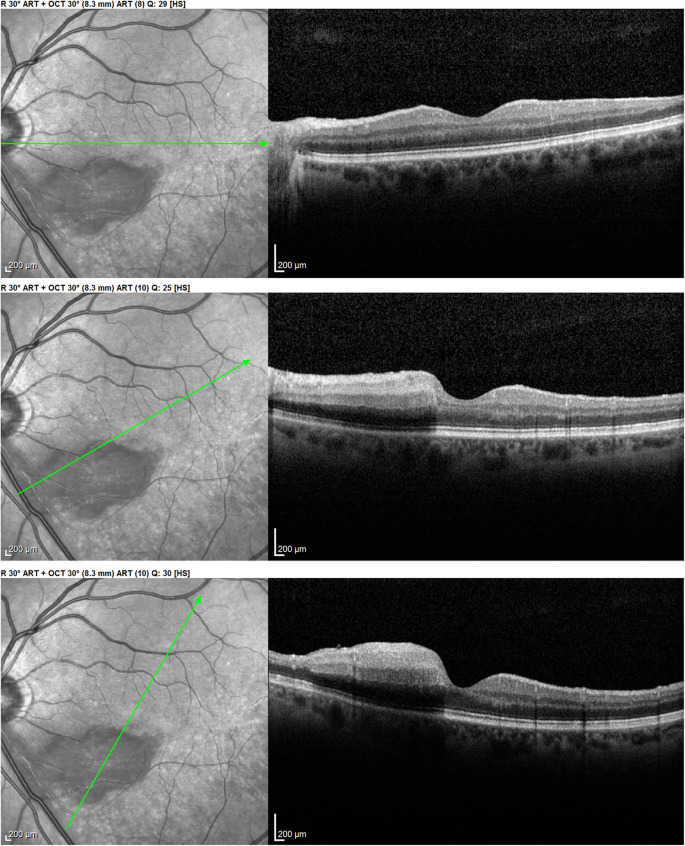
Optical Coherence Tomography (Oct) shows oedematous retinal thickening in the infarcted area in a patient seen in the acute phase (case 1) The SLO fundus prototype clearly delineates the ischaemic area
All other 3 patients showed areas of atrophic retinal thinning. (Fig. iv a & b)).
a OCT imaging of the retina in a patient iv months after retinal infarct showing atrophic thinning of the inner retina with conservation of the photoreceptor outer segments ellipsoid zone; notation white form of involved artery on the SLO fundus picture (case 3). b Microperimetry conspicuously delineating the not-functional atrophic retinal area (case 3)
Angiography
Angiographic signs were the most characteristic and relevant features that could be determined. In all four cases an abrupt stop or segmental interruption of flow was plant (Fig. 5, Fig. 6). The second sign was hyperfluorescence of the afflicted arteries present in all cases (Fig. 7). These two signs were more than clearly visible on ICGA in the three patients for whom this investigative process was performed (Table ii).
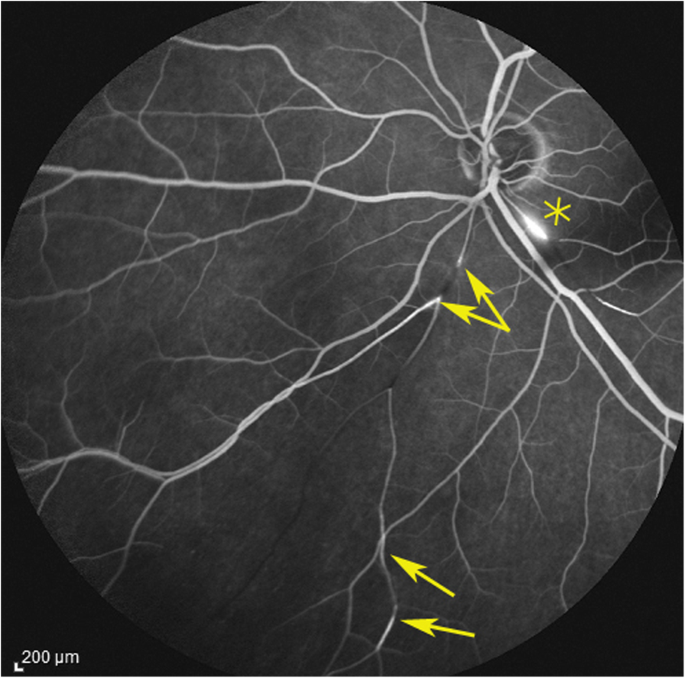
Fluorescein angiography (FA) showing abrupt intermission of two arteries inferiorly (arrows) and characteristic arterial wall hyperfluorescence (asterisk) (instance 1)
Indocyanine green angiography. Aforementioned view of fundus as on Fig. 5, showing precisely arterial end and/or interruption (arrows) (case 1)
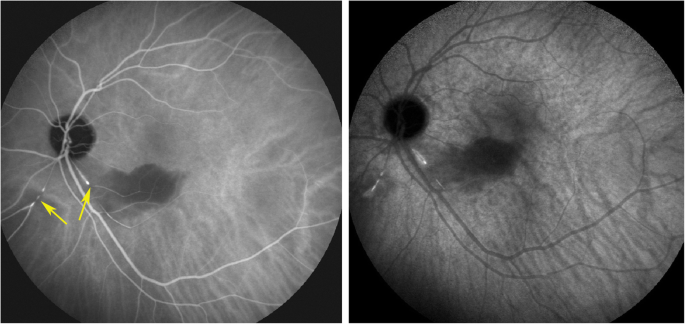
Fluorescein angiography (FA) showing segmental avenue occlusion and hyperfluorescence of the arterial walls in the left eye (ii correct pictures) in early (superlative) and late (bottom) angiographic phases. In the correct eye (two left pictures) an expanse of non-perfusion (capillary drop) of vessels in the mid-periphery (top) with staining of arteries meliorate seen in the late phase (bottom). (Case two)
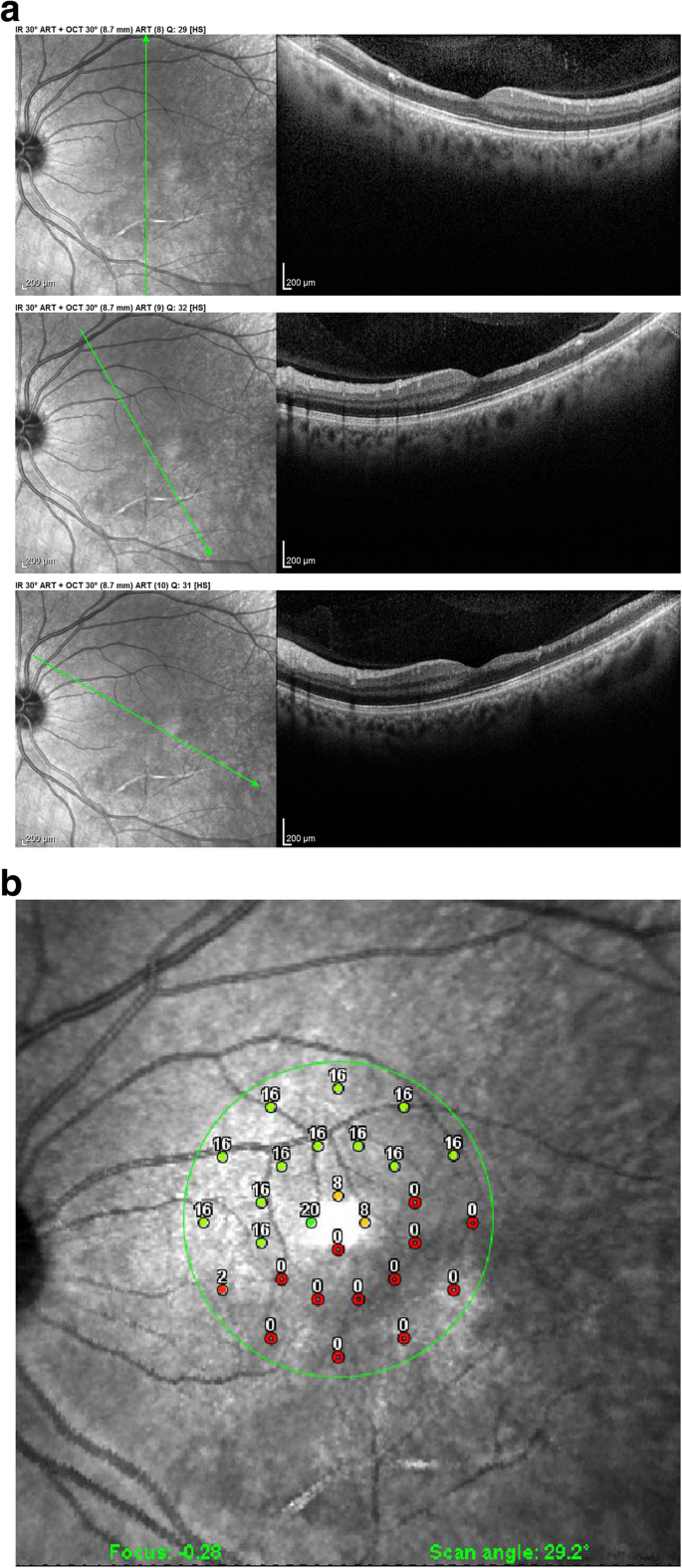
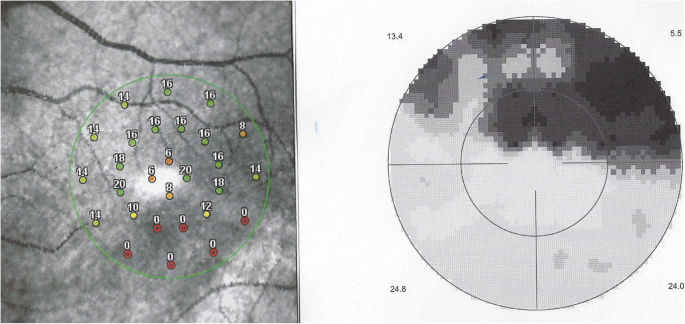
Scotomas were recorded in all four cases, corresponding to the areas of Retinal atrophy shown by October. (Fig. 8) with more precise delineation obtained by microperimetry. (Fig. 9).
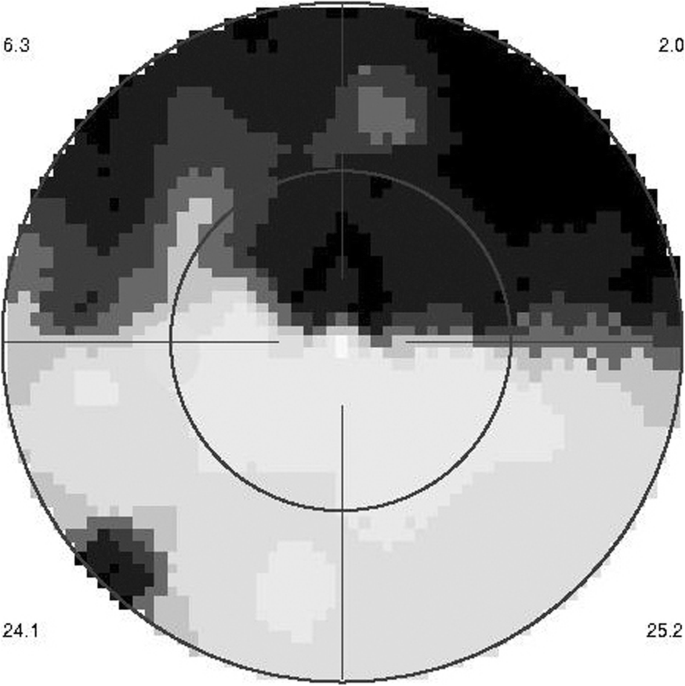
Vast upper scotoma corresponding to the ischaemic infarcted lower retina delineated by microperimetry (Fig. nine) (example i)
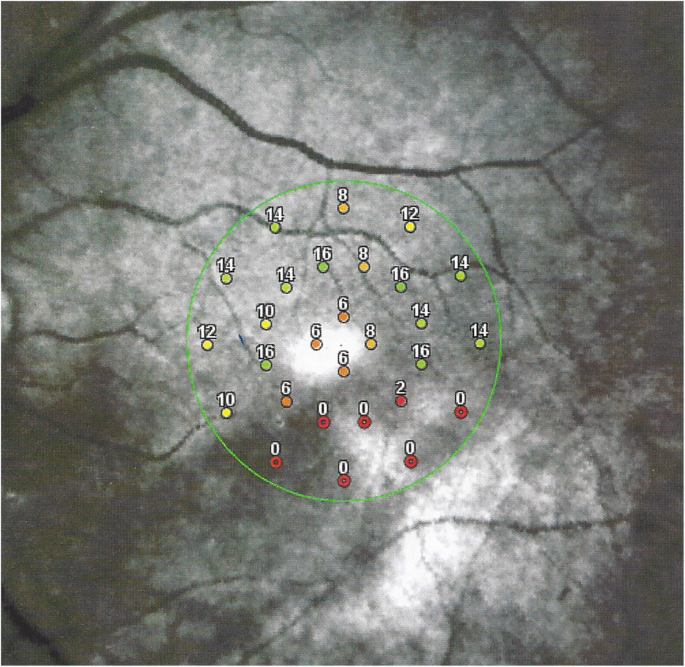
Microperimetry showing the area of functional impairment acquired by the retinal infarction. (Instance 1)
Associated investigations
MRI was negative in all four cases and a hearing loss was objectivised in 2 cases.
Handling
Patients 3 and four were under depression doses of systemic steroids at presentation. Patient 1 was treated with intravenous steroids (methylprednisolone 500 mg) for 3 days with a per os relay of steroids combined with mycophenolic acid (Myfortic®,1440 mg daily). Treatment was maintained and tapered over a period of 5 years. She is at present off handling without recurrence for nine months. Patient 2 was treated with 500 mg of intravenous methylprednisolone for 3 days followed past a combination of oral steroids and mycophenolate mofetil (Cellcept®, 2 grand daily) tapered afterwards a period of five years to twice 500 mg for ix months and and so discontinued The patient did not present a recurrence afterward a follow-up of vi years under treatment.
Patients three and 4 were both treated with 500 mg of intravenous methylprednisolone for 3 days associated with cyclosporine (Sandimmun 4 mg/kg) and mycophenolate mofetil (Cellcept®, 2 g daily) tapered over one–2 years. These 2 patients were lost to follow-up.
All four patients received anti-platelet handling, in form of aspirin 100–300 mg daily.
Evolution
Evolution was favourable in the two commencement patients who were devoid of recurrence after withdrawal of all treatment. Evolution was favourable in patients 3 and 4 just were subsequently lost for follow-up.
Case report
This 63 year old female patient woke-up ane morning with a large black dot which she described every bit a cloud in her central visual field, accompanied by scintillations. The patient mentioned hearing difficulties on her left side and a tendency to take retentivity blackouts since several weeks. Except for frequent migraine episodes without ocular symptoms her history did not reveal any particular boosted health problem. She consulted her center doctor who objectivised the scotoma by Goldman perimetry and who immediately referred the patient. Best corrected visual acuity was ane.0 (OD) and 0.ix (OS). At that place was no inductive segment inflammation, verified by laser flare photometry, and in that location were no cells in the vitreous. Fundus test showed an expanse of whitening of the retina situated inferiorly and nasally to the fovea. (Fig. 10) Octopus® (Haag-Streit), Bern, Switzerland) visual field testing showed superior-temporally to the fovea an accented scotoma. (Fig. 8) and microperimetry showed an absolute and relative loss of sensitivity of the retina corresponding to the whitish area seen on the fundus photography. (Fig. 9) Fluorescein angiography (FA) showed a perfusion delay in the lower half of the retina. The hitting finding however was segmental vasculitis of the arteries with in one expanse abrupt sub-apoplexy of an artery with intense bulging exudation. (Fig. five) On Indocyanine greenish angiography (ICGA) the segments of arteritis appeared besides hyperfluorescent. In add-on, ICGA displayed also hypofluorescence in the zone of retinal infarction that was either produced by the retinal edema and/or choriocapillaris circulation impairment due to the edema pressing on the choriocapillaris, or both. (Figs. 6 & eleven). Optical coherence tomography showed a substantial retinal edema caused by retinal infarction. (Figs. 3 & 12) The neurologic examination also as the cerebral MRI were inside normal limits. Intravenous methylprednisolone was immediately introduced (500 mg per 24-hour interval for iii days) followed by oral prednisone (sixty mg per 24-hour interval) associated with mycophenolic acid (Myfortic®, 1440 mg daily).and acetylsalicylic acid (300 mg daily). Later 5 weeks FA and ICGA findings reverted to normal with no arteritis and fading of the yellowish retinal infarcted area. (Figs. 13 and 14) Substantial thinning of the inner retina with conservation of the outer retina was seen on the follow-up OCT. (Fig. 12, bottom frame) Visual field and microperimetry showed a slight improvement. (Fig. 15).
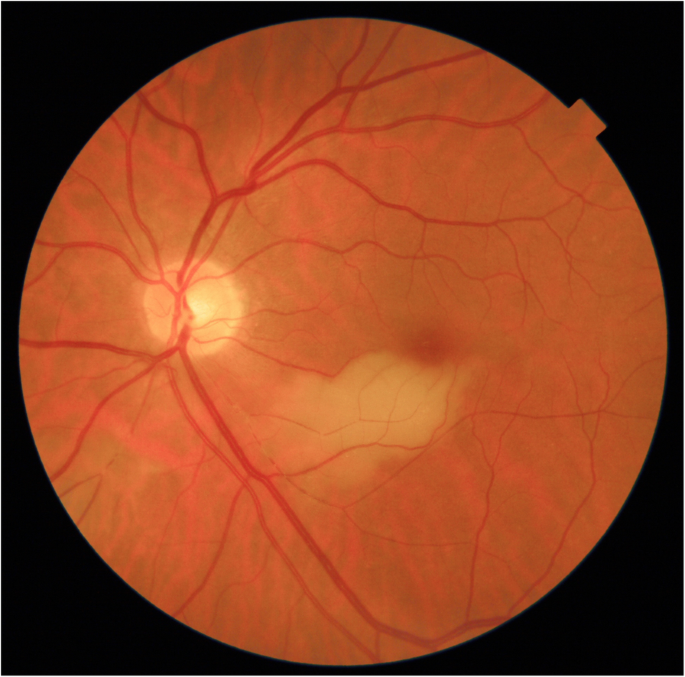
Yellow-white zone of retinal infarction (case 1)
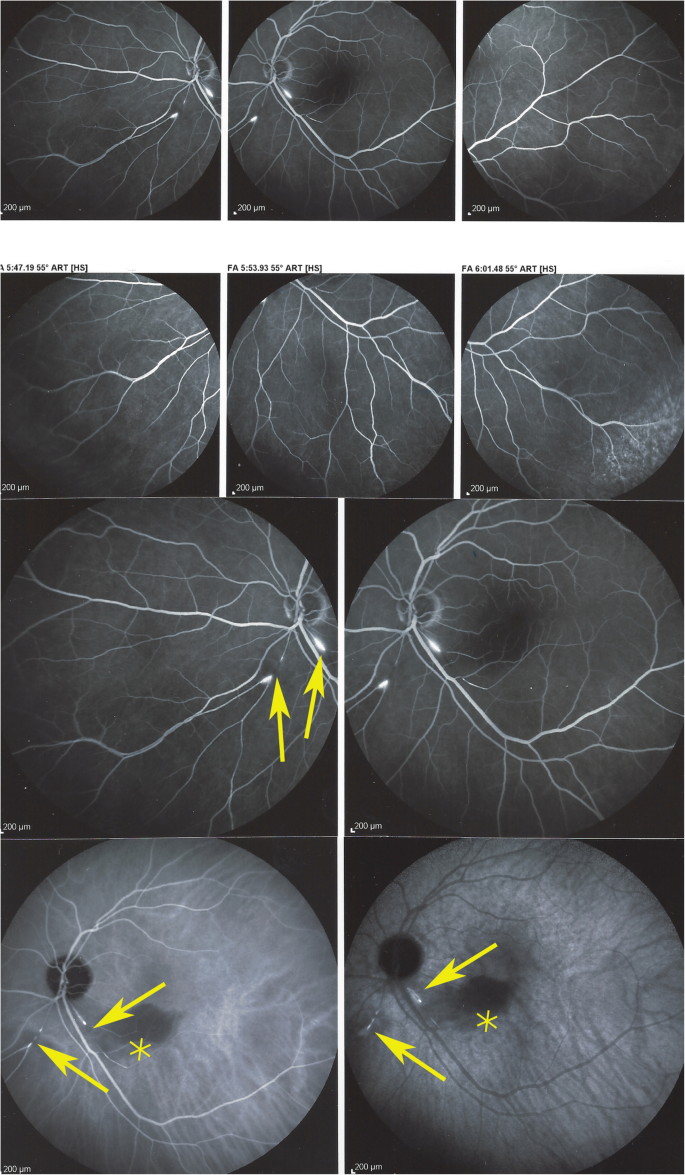
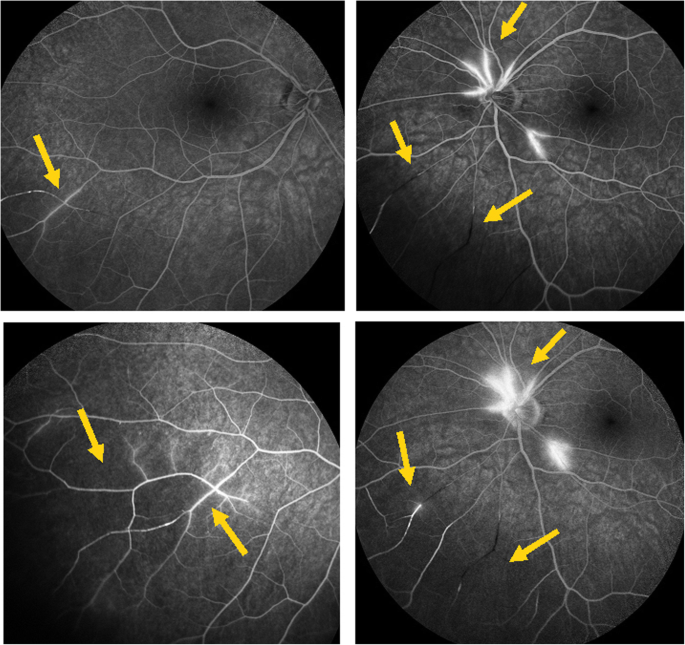
Panorama and posterior pole FA views showing arteritis with segmental involvement and abrupt artery occlusion or subocclusion (top sextet of frames). Posterior pole FA and ICGA views showing hyperfluorescence of the arteries on both FA (top two frames) and ICGA (bottom two frames) (arrows) and hypofluorescence in the areas of retinal infarction (asterisk) (bottom quartet of frames) (example 1)
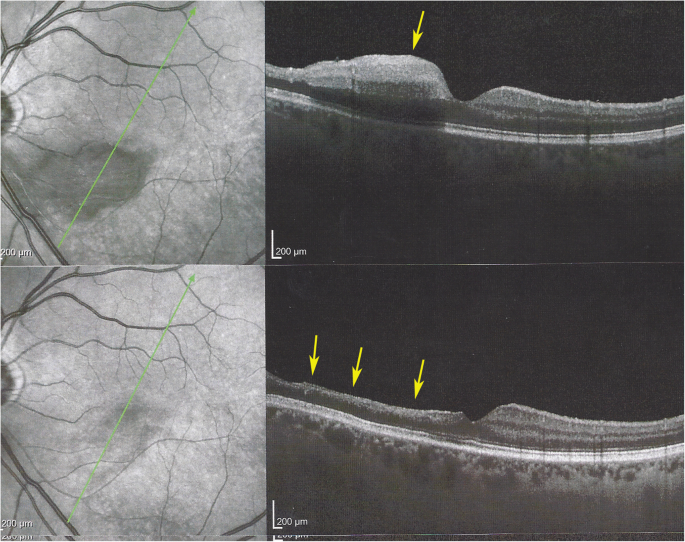
Evolution of OCT views of infarcted zone in the acute phase (top) showing retinal oedema (arrow) and 6 months later (lesser) showing atrophy of inner retina (arrow) while external retina is conserved. (case 1)
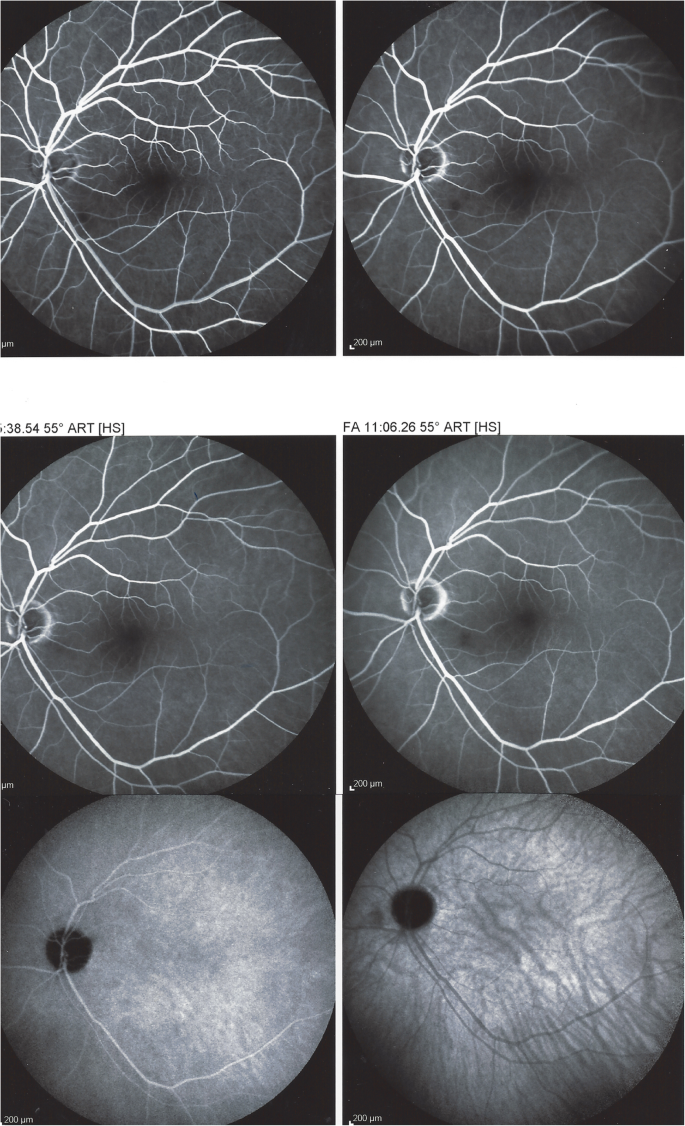
Follow-up FA (height iv frames) and ICGA bottom twoframes v weeks afterward acute episode and systemic corticosteroid treatment; complete resolution of arteritis (instance i)
Fundus picture OS taken 5 weeks after the astute episode showing quasi disappearance of whitish aspect in the infarcted zone (case 1)
Visual field and microperimetry six months after the astute episode, showing improved microperimetry score (left) and slight reduction of scotoma (correct) (case 1)
Patient i was treated with intravenous steroids (methylprednisolone 500 mg) for 3 days with a per os relay of steroids combined with mycophenolic acrid (Myfortic®,1440 mg daily). Treatment was maintained and tapered over a period of 5 years. Off treatment for ix months of follow-up she did not show a recurrence.
Word-determination
Susac syndrome is a vasculitis of rare occurrence making up for 0.xv% of cases in a uveitis referral middle. In all our patients, it was the ocular involvement that atomic number 82 the patients to consult the ophthalmologist whose responsibility it is to perform a prompt diagnosis in guild to avoid cerebral and cochlear complications if they are not yet nowadays. Our findings indicated that the diagnosis is well managed in uveitis referral centres simply is missed by ophthalmologists at large.
We identified potentially illness defining findings thanks to angiographic investigation. In all patients we found constant features on fluorescein angiography consisting in precipitous and/or segmental arterial stops likewise every bit sectorial hyperfluorescent arterial vessel walls autonomously from the occluded arteries. These FA signs, typical for SS were also seen on ICGA which identified these lesions more precisely. As reported earlier, we too establish that ICGA showed no circulatory problems/ischemia at the level of the choroid, except a hypofluorescence in the area of the retinal infarct. We propose to survey such findings in order to orient patients from the unspecific diagnosis of vasculitis towards the diagnosis of SS. In these cases that present ocular interest offset, the ophthalmologist's office is crucial and acts as a whistle blower, by recognising and treating the patients and so avoiding astringent cochlear and cognitive complications. Rapid identification of the illness by the ophthalmologist is crucial, as in ophthalmological series, unlike in neurological series, MRI is often negative and ophthalmic manifestations may exist the sole presenting sign [29,30,31,32]. This is one more reason to take good ophthalmological disease defining criteria. In our experience all cases responded well to prompt dual steroidal and non-steroidal immunosuppression of more than 1–5 years' elapsing and cerebral involvement was prevented by therapy initiated promptly in three patients.
Diagnosis of SS is primarily based on the clinical presentation, in particular ocular angiographic signs associated with auditory and central nervous organization findings forming the classical triad. Susac syndrome is often misdiagnosed or diagnosed very late in the clinical course. I reason is the fact that the consummate clinical triad is present in only about xv% at the onset of the disease [17]. The average delay betwixt the kickoff symptoms and the complete triad can range from some weeks to more than than 2 years [4, 6]. In our patients, the time menstruum betwixt the disease onset and the correct clinical diagnosis was from 1 calendar week to 126 months, as reported by other groups [6].
Susac syndrome is presumed to be an immune-mediated microangiopathy (endotheliopathy) of the retina, the cochlea and the brain. Information technology is characterized by a clinical triad of branch retinal artery apoplexy, hearing loss and encephalopathy. The handling has to exist early, aggressive and long enough, combining steroidal and non-steroidal immunosuppression to avert visual loss, deafness and dementia.
Availability of data and materials
The information used during the electric current article are available from the corresponding author on reasonable request.
References
-
Papo T, Biousse V, LeHoang P, et al. Susac syndrome. Medicine (Baltimore) 77:3–11
-
Susac JO, Hardman JM, Selhorst JB. Microangiopathy of the encephalon and retina. Neuroloogy 1979; 29: 313–316
-
Susac JO (1994) Susac'southward syndrome: the triad of microangiopathy of the brain and retina with hearing loss in young women. Neurology 44:591–593
-
Dörr J, Krautwald S, Wildemann B et al (2013) Characteristics of Susac syndrome: a review of all reported cases. Nat Rev Neurol ix:307–316
-
Susac JO, Egan RA, Rennebohm RM, Lubow One thousand (2007) Susac's syndrome: 1975-2005 microangiopathy/autoimmune endotheliopathy. J Neurol Sci 257:270–272
-
Jarius S, Kleffner I, Dörr JM et al (2014) Clinical, paraclinical and serological findings in Susac syndrome: an international multicenter study. J Neuroinflammation 11:46
-
Magro CM, Poe JC, Lubow Grand, Susac JO (2011) Susac syndrome: an organ-specific autoimmune endotheliopathy syndrome associated with anti-endothelial prison cell antibodies. Am J Clin Pathol 136:903–912
-
Gross CC, and CD8+ T-cell-mediated endotheliopathy is a targetable machinery of neuro-inflammation in Susac syndrome. Nat Commun 2019:; 10 (1) 5779. Doi: https://doi.org/ten.1038/s41467-019-13593-five
-
McLeod DS, Ying HS, McLeod CA, Grebe R, Lubow M, Susac JO, Lutty GA (2011) Retinal and optic nerve head pathology in Susac'south syndrome. Ophthalmology 118:548–552
-
Egan RA, Hills WL, Susac JO (2010) Gass plaques and fluorescein leakage in Susac syndrome. J Neurol Sci 299:97–100
-
Egan RA, Ha Nguyen T, Gass JDM, Rizzo JF, Tivnan J, Susac JO (2003) Retinal arterial wall plaques in Susac syndrome. Am J Ophthalmol 135:483–486
-
Brandt AU, Zimmermann H, Kaufhold F, et al. Patterns of retinal damage facilitate differential diagnosis between Susac syndrome and MS. (Schuelke M, Ed.) PLoS One seven: e38741
-
Martinet N, Fardeau C, Adam R, Bodaghi B, Papo T, Piette JC, Lehoang P (2007) Fluorescein and indocyanine green agiographies in Susac sydrome. Retina 27:1238–1242
-
Ringelstein M, Albrecht P, Kleffner I et al (2015) Retinal pathology in Susac syndrome detected by spectral-domain optical coherence tomography. Neurology 85:610–618
-
Susac JO, Murtagh FR, Egan RA, Berger R, Lincoff N, Gean Advertising, Galetta SL, Fob RJ, Costello FE, Lee AG, Clark J, Layzer RB, Daroff RB (2003) MRI findings in Susac's syndrome. Neurology 61:1783–1787
-
Dörr J, Ringelstein M (2014) Duning T & Kleffner I update on Susac syndrome: new insights in brain and retinal imaging and handling options. J Alzheimers Dis 42(Suppl three):S99–S108
-
Kleffner I, Duning T, Lohmann H et al (2012) A brief review of Susac syndrome. J Neurol Sci 322:35–40
-
Rennebohm R, Susac JO, Egan RA, Daroff RB (2010) Susac's syndrome--update. J Neurol Sci 299:86–91
-
Petty GW, Mattson EL, Younge BR, McDonald T, Forest CP (2001) Recurrence of Susac syndrome (retinochleocerebral vasculopathy) subsequently remission of 18 years. Mayo Clin Proc 76(9):958–960
-
Buzzard KA, Reddel SW, Yiannikas C, Sean Riminton D, Barnett MH, Hardy TA (2015) Distinguishing Susac'south syndrome from multiple sclerosis. J Neurol 262:1613–1621
-
Rennebohm RM, Susac JO (2007) Treatment of Susac'south syndrome. J Neurol Sci 257:215–220
-
Rennenbohm RM, Egan RA, Susac JO treatment of Susac's syndrome Curr Treat Options Neurol (2008) 10: 67–74
-
Mateen FJ, Zubkov AY, Muralidharan R, Fugate JE, Rodriguez FJ, Winters JL, Petty GW (2012) Susac syndrome: clinical characteristics and treament in 29 new cases. Eur J Neurol 19:800–811
-
Greco A, De Virgilio A, Gallo A, Fusconi G, Turchetta R, Tombolini 1000, Rizzo MI, de Vincentiis G (2014) Susac's syndrome--pathogenesis, clinical variants and treatment approaches. Autoimmun Rev 13:814–821
-
Rennebohm RM, Lubow M, Rusin J, Martin L, Grzybowski DM, Susac JO. Aggressive immunosuppressive treatment of Susac's syndrome in an adolescent: using treatment of dermatomyositis as a model. Pediatr Rheumatol Online J 2008; 29;half dozen:3 doi: https://doi.org/10.1186/1546—0096-half dozen-three
-
Kleffner I, Dörr J, Duning T, Immature P, Ringelstein EB, Schilling M (2014) Susac syndrome treated with subcutaneous immunoglobulin. Eur Neurol 71:89–92
-
Hardy TA, Garsia RJ, Halmagyi GM, Lewis SJG, Harrisberg B, Fulham MJ, Barnett MH (2011) Neoplasm necrosis factor (TNF) inhibitor therapy in Susac's syndrome. J Neurol Sci 302:126–128
-
Buelens T, Ossewaarde-van Norel J, de Boer JH, Nurbourgh I, Gilbert Grand, Kamgang Semeu P, Fils JF, Caspewrs L, Posrtelmans L, Willermain F (2020) Evaluation of tumor necrosis gene inhibitor therapy in Susac syndrome. Retina 40:581–590
-
Apostolos-Pereira SL, Passos Kara-José LB, Euripedes Marchiori P, Ribeiro Monteiro ML (2013) Unilateral central retinal artery apoplexy as the sole presenting sign of Susac syndrome in a boyfriend: case report. Arq Bras Oftalmol 76:192–194
-
Garcia-Basterra I, Baquero Aranda I, Garcia-Ben A, Garcia-Campos JM (2018) Susac's syndrome : isolated retinal artery occlusion after 10 years of remission. Arq Bras Oftalmol 81:144–147
-
Snyers B, Boschi A, De Potter P, Duprez T, Sindic C (2006) Susac syndrome in 4 male patients. Retina 26:1049–1055
-
Sauma J, Rivera D, Wu A, Donate-Lopez J, Gallego-Pinazo R, Chilov Chiliad, Wu M, Lihteh West (2020) Susac'southward swyndrome: an update. Br J Ophthalmol 104:1190–1195
Acknowledgements
Not applicable.
Writer information
Affiliations
Contributions
IP contribute in acquisition, analyse and interpretation of the data. He contributed in writing and reviewing of the manuscript. BT has contributed in writing, reviewing and collecting the information of one example. CHP was the designer of the written report and was the major reviewer of the manuscript. He also contributed in writing. All authors read and approved the concluding manuscript.
Authors' information
Not applicable.
Respective writer
Ethics declarations
Ethics blessing and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
In that location is no financial disclosure.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open up Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, accommodation, distribution and reproduction in any medium or format, as long every bit yous give advisable credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third political party cloth in this commodity are included in the commodity'southward Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, y'all will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
Reprints and Permissions
About this article
Cite this article
Papasavvas, I., Teuchner, B. & Herbort, C.P. Susac syndrome (Retino-cochleo-cerebral vasculitis), the ophthalmologist in the office of the whistleblower. J Ophthal Inflamm Infect ten, 27 (2020). https://doi.org/10.1186/s12348-020-00217-z
-
Received:
-
Accepted:
-
Published:
-
DOI : https://doi.org/10.1186/s12348-020-00217-z
Keywords
- Susac syndrome
- Branch retinal artery occlusion (BRAO)
- Fluorescein angiography
- Indocyanine green angiography
Source: https://joii-journal.springeropen.com/articles/10.1186/s12348-020-00217-z
0 Response to "A Review and Update on the Ophthalmic Implications of Susac Syndrome"
Post a Comment